Newsroom
The latest news and updates on Sooma tDCS™, neurostimulation treatment, and what’s happening at Sooma Medical.


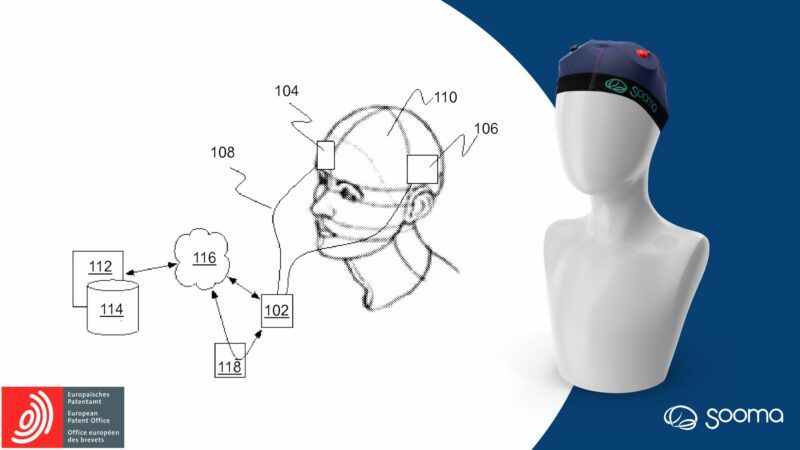
FDA Breakthrough Device Designation granted to Sooma for Its Innovative At-Home Depression Treatment Device
Read more
New European Regulation reclassifies brain stimulation equipment without an intended medical purpose as class III
Read more
Sooma’s latest study provides the largest dataset to date on the effects of tDCS for treating MDD in real-world clinical practice
Read more
Sooma reaches more than 10,000 patients treated with its tDCS devices
Read more
Sooma starts a collaboration with the Non-Invasive Brain Stimulation Lab at University Medical Center (Göttingen)
Read more
Treating depression shouldn’t be hard
Read more
tDCS therapies are now reimbursed in Estonia
Read more
Time to take action on mental health issues
Read more
Solving the most common pitfalls of tDCS therapies
Read more
Sooma welcomes a new Scientific Board Member
Read more