tDCS therapies are now reimbursed in Estonia

Psychiatric and neuropathic patients in Estonia can now access tDCS treatments more efficiently, as the Estonian public healthcare system is currently reimbursing these neuromodulation therapies as part of its service portfolio.
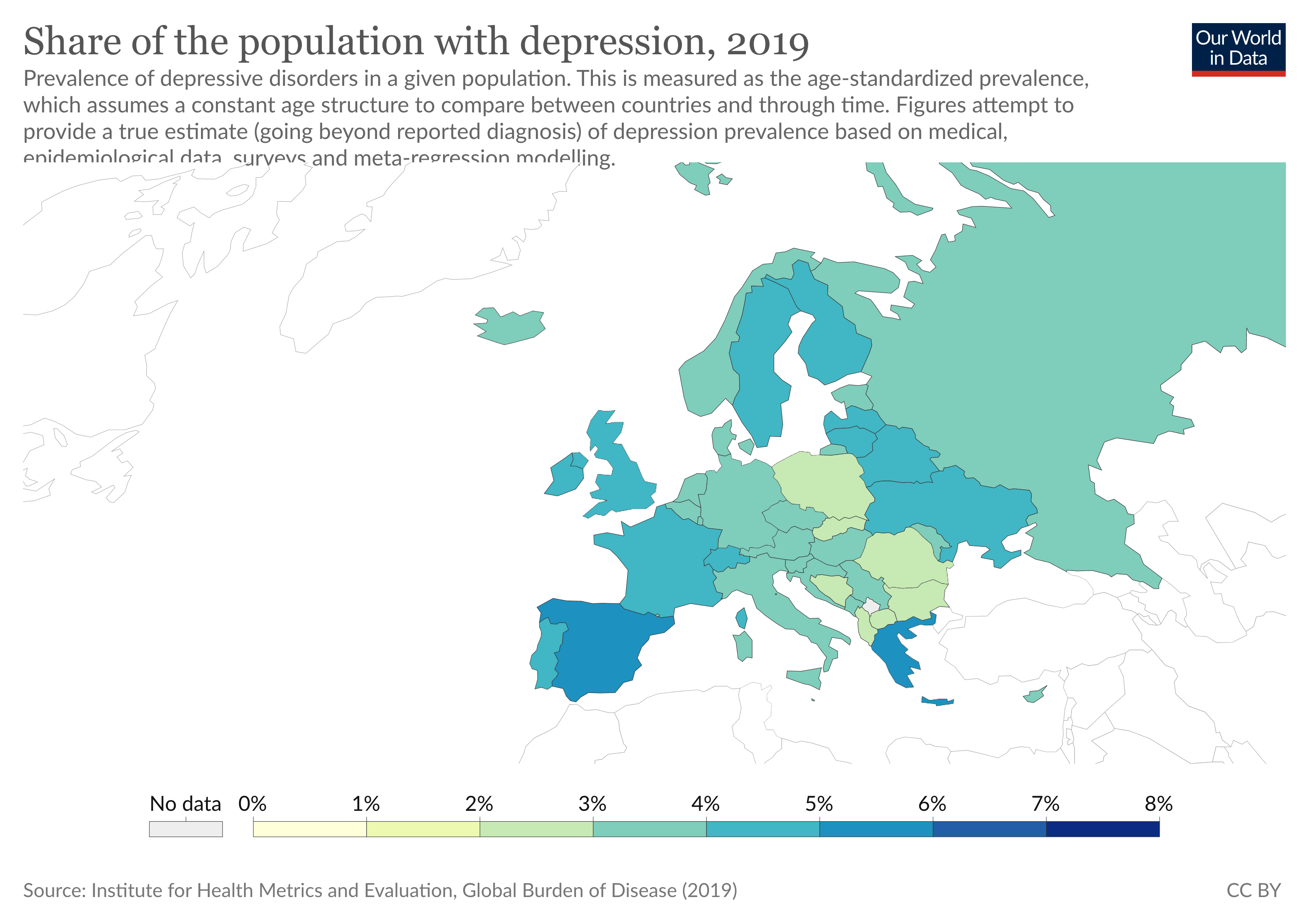
With 1.3 million people, Estonia has a depression rate of about 4% (Our World in Data, 2019). After this decision, it has become the second country in Europe, after Finland, to officially support tDCS therapies as an effective tool to treat a wide range of ailments, such as depression and chronic pain.
More than ten public hospitals and private clinics offer Sooma tDCS treatments in their portfolio, including leading area hospitals and national-level clinics. But, more importantly, now healthcare professionals all around the country have one more safe, effective, and accessible treatment option to offer their patients with psychiatric or neurologic disorders.
TDCS is the treatment method offered by Sooma for depression (Sooma Depression Therapy, indicated for Major Depressive Disorder) and chronic pain (Sooma Pain Therapy, indicated for Fibromyalgia and chronic neuropathic pain). By using Sooma devices, you ensure that you are performing a safe and effective patient treatment, should be it in the hospital or at patients’ homes, with legal, regulated, tested, and effective equipment that complies with the latest EU regulations and features all the recommended elements listed on this article.
Latest news

Sooma Announces Medical Technology Expert Andreas Hartlep as New CEO
Read more
TGA approves Sooma’s at-home brain stimulation for depression in Australia
Read more
Sooma Medical Announces Pivotal FDA IDE Clinical Trial for At-Home Brain Stimulation Device for Depression Treatment
Read more